SOAPS, SANITIZERS, and DISINFECTANTS
How to Effectively Prevent COVID-19 Infections
What we know as of April of 2020, three of the most effective ways to prevent COVID-19 infections are:
- hand washing with soap and water.
- the use of hand sanitizers when washing with soap and water is not available.
- the use of disinfectants on surfaces in our homes and businesses.
SOAPS
All the information we have received thus far about the
COVID-19 (coronavirus) stresses that washing hands with soap and water
regularly is one of the most effective ways of preventing infection.
WHY HAND WASHING WITH SOAP and WATER IS SO IMPORTANT
In the case of viruses, the entry into the body is through the mouth, nose, or eyes, and that is most likely to happen by touching the face with contaminated hands. Soapy water washes away microbes very effectively from hands, better than just water. That is because our skin is coated with sebum, a mixture of fatty materials that prevent dryness by inhibiting the evaporation of moisture from the skin.
Viruses can embed themselves in this fatty layer. Since water and fat do not mix, rinsing with just water is ineffective. But soap is what is called a ‘surfactant.’ This is a molecule that has a water-soluble end and an oil-soluble end. One end attracts oil, the other water, and this allows oily deposits to be rinsed away.
Coronaviruses have a protective coating of proteins and fats. Because of this ability to ‘wash away’ fatty materials, washing with soap and water also washes away the fat layer that protects the coronavirus. This will effectively kill the virus. As written by Dr. Joe Schwarcz on the McGill University Office for Science and society website:
"In the case of COVID-19, soap can actually destroy the virus responsible for the disease. Coronaviruses are composed of a core of nucleic acids, either RNA or DNA, surrounded by a protective coating made of proteins and fats. The fat-soluble end of the soap molecule embeds itself in the lipid layer and the virus is then literally pulled apart since the rinsing water is tugging the other end. The reason for the 20-30 second time period is to ensure that the soap makes contact with whatever microbes may be present."
WHICH IS MORE EFFECTIVE?
LIQUID SOAP or BAR SOAP
LIQUID SOAP
Liquid soap is not actually a soap but a ‘synthetic detergent’ that was originally developed for laundry purposes. As far as cleaning action goes, it doesn’t make any difference whether you use a liquid or bar soap. But from an environmental point of view bar soap wins. Liquid soap has a carbon footprint about 25% greater than that of bar soap.
LIQUID SOAP DOWNSIDES
- Liquid soap requires a plastic bottle that takes more energy to produce than the wrapping of a bar of soap.
- The container of liquid soap is usually made of recyclable (yet often doesn’t get recycled) high-density polyethylene (HDPE) which requires petroleum or natural gas for production.
- Water makes up most of liquid soap, making it heavier and requiring more energy to transport.
- On average, people use about 2.3 grams of liquid soap to wash their hands but only about .35 grams of a bar soap. This means, of course, the greater use of raw materials, more processing, and more carbon dioxide emissions.
- Liquid soap also costs about ten times as much to wash your hands than bar soap. Even bar soap harboring bacteria will not leave a residue on hands. All the bacteria will be washed away with the soap.
BAR SOAP DOWNSIDES
- More hot water is used with bar soap.
- The raw material in the making of bar soap is usually a vegetable fat, such as palm or olive oil (Palmolive), so that agricultural chemicals and land use have to be taken into account.
Below is a video from the World Health Organization showing the proper way to wash hands to prevent COVID-19 infection:
DRYING AFTER WASHING HANDS IS AS CRITICAL AS WASHING FREQUENTLY!
We know that whether you use bar or liquid soap, the important thing is to wash hands frequently! Just as important is to dry them well after washing! That is critical because moist hands are much more likely to pick up microbes from a surface. Although electric hand driers can be generally effective, people don’t use them long enough to dry their hands completely.
Paper towels are a much better option. Not only because they can dry your hands better but you can also then use them to close the faucets and touch the doorknob just in case the person before you had failed to practice safe hand washing techniques.
HAND SANITIZERS
Washing with soap and water, as mentioned above, is more effective than using an alcohol-based hand sanitizer. That is because alcohol (at least 60%) can also dissolve fats and can strip away the lipid or fat layer of a virus, inactivating it.
Just using sanitizer without washing with soap and water is not recommended because sanitizer does not remove dirt and may therefore not be able to reach viruses that may be stuck in the dirt. There is no question, though, that when soap and water are not available, hand sanitizers can be useful.
There is actually a proper way to use sanitizer to get maximum effectiveness from the product:
- Before you start, make sure your hands are clean of visible dirt and
grime.
- Using about a palm-sized amount of the sanitizer, rub your hands briskly together for 20 to 30 seconds, taking care to get underneath your fingernails, between your fingers, the back of your hands and your wrists.
- Allow the sanitizer to dry completely, without wiping your hands off or rinsing them with water.
- Once the sanitizer dries completely, the process is complete.
DO HAND SANITIZERS CONTAIN INGREDIENTS HARMFUL TO DOGS?
Have you seen the 'warning' that hand sanitizers may have the same ingredients as antifreeze and are therefore toxic to dogs? Have you been afraid to let your animals lick your hands after using sanitizer?
The 'warning' is based on a misunderstanding of the difference between the chemicals ethylene glycol and propylene glycol and is not factual:
Ethylene Glycol
Ethylene gloycol is the usual active ingredient in antifreeze and is not only toxic to dogs but to humans as well.
Propylene Glycol
Propylene Glycol has an extra carbon atom and is used in sanitizers to hold in moisture in order to prevent hands from drying out. It is also found in some antifreeeze, especially those promoted as having a low toxicity and being environmentally friendly because it replaces the ethylene glycol, the usual active ingredient in antifreeze.
HOW TO MAKE YOUR OWN HAND SANITIZER
Recipes for hand sanitizer have been flooding social media. This one is from My Heavenly Recipes:
INGREDIENTS:
- 2/3 cup 99%* Rubbing Alcohol
- 1/3 cup Aloe Vera Gel
- Essential Oil of Your Choice (optional).
DIRECTIONS:
- Put the rubbing alcohol and aloe vera gel into a small bowl.
- Mix well.
- Add a few drops of an essential oil if you would like. Place in containers with lids or pumps.
*NOTE: In order to be effective, hand sanitizers must be at least 60% alcohol. When you dilute with Aloe Vera, the alcohol content is cut by 33%. If you start with 90% alcohol, this recipe will make one cup of sanitizer with 65% alcohol.
DISINFECTANTS TO USE WHEN CLEANING SURFACES
The key ingredients to look for in commercial sprays are hydrogen peroxide or calcium hypochlorite which have been shown to be effective against viruses.
Commercial wipes should be treated with quaternary ammonium salts such as benzalkonium chloride or benzethonium chloride in order to be effective against viruses.
IMPORTANT: AFTER SPRAYING SURFACES WITH DISINFECTANTS, DO NOT DRY IMMEDIATELY
It is important to allow surfaces that have been sprayed with disinfectants to be in contact with the surface before being dried. This is because contact time is needed to inactivate the virus on surfaces.
HOW TO MAKE DISINFECTING WIPES AT HOME
The following recipe is from My Heavenly Recipes:
INGREDIENTS:
- Roll of Paper Towels
- 2 cups Distilled water
- 1 cup Isopropyl alcohol- at least 70-90% concentration
- 1 TBSP Dawn dish soap (not Ultra)
- 3 drops Tea tree oil (Optional)
DIRECTIONS:
- Cut the paper towel roll through the middle with a serrated knife. Put in a jar or previous Lysol/Clorox wipes box.
- Mix together the water, rubbing alcohol, and Dawn dish soap.
- Pour around the edges of the paper towel roll and let it soak in for 2 minutes. Throw out the cardboard from the middle of the roll so you can pull a piece from the idle. Store in a closed container with a lid.
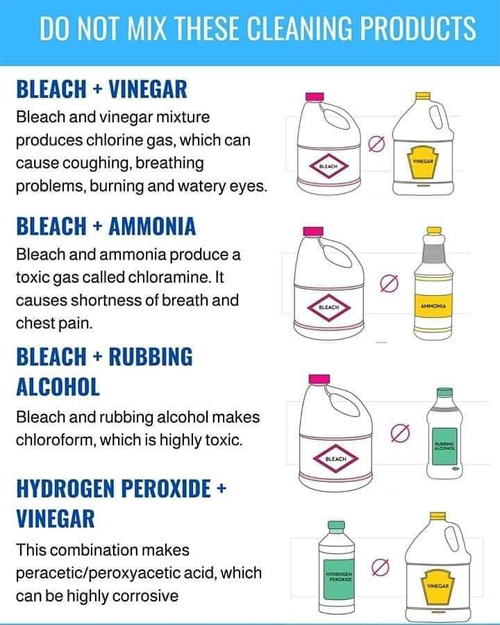
Please adhere to the advice on the following chart when making any DIY cleaning products:
HOW ABC CAN HELP
We can thoroughly clean and sanitize your carpets, furniture, tile and grout, and area rugs and offer information on proper cleaning techniques.
During the COVID-19 pandemic, however, we will be suspending our cleaning in occupied homes for the safety of our customers as well as our employees. We will continue to clean vacant apartments, homes, and businesses.
We will also continue to offer our EPA-registered sanitizer at no extra charge for the carpet, upholstery, and tile and grout surfaces in these buildings.
For oriental and area rugs, we have developed a 'no-contact' policy for pickups and deliveries as well as for customers bringing these items to our rug plant for cleaning and sanitizing.
Please also see our articles on 10 Favorite Cleaning Hacks, Green Cleaning Solutions, Green Cleaning Options, among others that can be found here.
You can always call or text our office at 607-272-1566 and visit our website for more information.
PLEASE NOTE: Much of our scientific information has been used with permission from Joe Schwarcz, Ph.D., at the McGill University Office for Science and Society.
Top of Soaps, Sanitiziers, and Disinfectants
"The Cleanest Clean You've Ever Seen."
by
ABC Oriental Rug & Carpet Cleaning Co.
130 Cecil Malone Drive Ithaca, NY 14850
607-272-1566