mRNA VACCINES
mRNA vaccines (messenger RNA) are the newest players in the race to defeat the COVID-19 pandemic that is currently engulfing our world (as of December 2020).
An mRNA vaccine is very different from a traditional vaccine. Many companies worldwide have been working on developing this type of vaccine specifically for the SARS-CoV-2 virus but none have yet (as of this writing) been approved.
But what are mRNA vaccines? How do they work? Are they effective? Are they safe? Can they be developed quickly and in large enough quantities for mass immunization? These are just a few of the questions that must be answered before any mRNA vaccines can be approved for distribution.
Here is what we know:
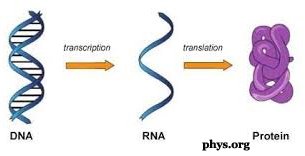
DNA & RNA in HUMANS & VIRUSES
DNA or deoxyribonucleic acid is the molecule that contains each organism's genetic code. In humans and other mammals, the DNA remains in the nucleus of each cell and the genetic instructions are copied into the RNA (ribonucleic acid), which in turn acts as a messenger and brings the copies to the rest of the cell through a process that forms the proteins that are needed to carry out our bodily functions.
However, in some viruses, like COVID-19, RNA rather than DNA carries the genetic information. It is possible to produce synthetic COVID-19 RNA molecules in a lab capable of provoking our immune system to form a defense strong enough to kill the actual virus should it invade our systems.
OUR IMMUNE SYSTEMS and VACCINES
Our immune systems are composed of 2 parts. The innate immune system is the immunity we are born with. The acquired immune system is that which develops as we come into contact with pathogens.
Traditional vaccines usually only work with the acquired immune system and need another ingredient, called an adjuvant to activate the innate immune system.
The mRNA
vaccine can activate the innate immune system as well as the acquired immune system without the need to add
adjuvants. The type of immune response from an mRNA vaccine would be a very strong one.
mRNA VACCINES for COVID-19
Most of what we know about mRNA or messenger RNA vaccines has come from work in the cancer field. Every cancer tumor is different. Tumor-specific mRNA vaccines have been developed to target an individual patient's tumors. They activate the body’s T-cells (the acquired immune system that kills cells) to destroy those tumors.
In viral infections such as the coronavirus, a strong T-cell response is needed, but with a virus such as SARS-CoV-2 (COVID-19), another part of the acquired immune system, the B-cells, need to be activated as well to produce antibodies that will mark the virus for destruction. The B-cells are not as relevant for cancer tumors so there has been limited experience with B-cell responses and mRNA vaccines.
TRADITIONAL VACCINES vs
mRNA VACCINES
Traditional vaccines use small or inactivated doses of either a whole disease-causing organism such as a virus or a bacteria or the proteins it produces. When introduced into the body, the vaccine trains the body to recognize and respond to the proteins of the virus or bacteria and provokes the immune system to mount an attack.
mRNA vaccines, on the other hand, actually trick the body into producing some of the proteins of the virus or bacteria itself. In order to produce an mRNA vaccine, a synthetic version of the actual RNA (the code that a particular virus uses to build its infectious proteins) is produced. When this RNA is delivered into the body, it is read as instructions to create some of the virus’s molecules. But, not to worry! These proteins are solitary and do not assemble to form a virus. The immune system will detect these viral proteins and start to produce a defensive response to them.
THE WORK CONTINUES FOR SAFE AND EFFECTIVE mRNA VACCINES
Scientists are working feverishly to understand the immune response to the mRNA vaccines, how long the protection they give would be, what, if any, are the downsides, as well as what would be the optimal route for vaccine delivery, such as needle-syringe injections, injections into blood, muscle, lymph node, directly into organs, or via a nasal spray, etc.
One positive advantage of mRNA vaccines is that by getting the human body to produce the virus’s proteins itself, some of the manufacturing process could be cut out making it easier and quicker to produce than traditional vaccines. Another major advantage is that mRNA can be produced in the laboratory using readily available materials, making it less expensive and faster than conventional vaccine production which may require the use of chicken eggs or other mammalian cells.
HUMAN CLINICAL TRIALS OF mRNA VACCINES
Moderna, a US company, began its first human trials of an mRNA vaccine in March of 2020. Recently, Pfizer and its German partner, BioNTech SE, stated that early analysis shows their COVID-19 mRNA vaccine is more than 90 per cent effective at preventing symptomatic illness.
But there is a lot we do not know about the human clinical trials any companies have used or are using. Since these might be the first mRNA vaccine to be used against the COVID-19 coronavirus in humans, it is important to look very carefully at all the data, especially the safety data. Further research is needed to address many technical difficulties such as vaccine stability and delivery. At this time, it is also not certain which production method(s) are the best or what quantities of mRNA will be sufficient to produce an adequate immune response in a human.
Although human clinical trial data is still limited and more long-term studies are needed to determine the effectiveness of mRNA vaccines, in the face of the continuing severity of the COVID-19 pandemic these companies may apply for emergency use authorization of their vaccines.
Pfizer will soon have 2
months’ worth of safety data. (Their vaccine requires 2 doses, a month apart
and no safety concerns have arisen as yet). Moderna is also planning to apply with its mRNA vaccine. With both of these vaccines, there is a concern because their stability requires they be stored in frozen form requiring special equipment for shipping and storage. Many other pharmaceutical companies are also working on mRNA vaccines as well as on other vaccine types.
All of this feverish vaccine activity means we may very soon see a welcome bit of light at the end of the COVID-19 tunnel. It is, at the very least, encouraging.
"The Cleanest Clean You've Ever Seen."
by
ABC Oriental Rug & Carpet Cleaning Co.
130 Cecil Malone Drive Ithaca, NY 14850
607-272-1566